Molar mass of carbon periodic table

Coal is the most common example of amorphous carbon. One of the most common of these is amorphous carbon which does not have a defined crystal structure. There are many different forms or allotropes of carbon. The element carbon continues to be actively investigated and plays a vital role in all fields of chemistry. Fullerene even won a Nobel Prize in Chemistry in 1996 the prize went to Robert Curl, Sir Harold Kroto, and Richard Smalley. Many other forms of carbon have been discovered such as diamonds, graphite, graphene, and fullerene. The use of carbon continued until 1789 when Antoine Lavoisier listed carbon as an element for the first time. As early as 3750 BC, ancient Egyptians and Sumerians used charcoal, a form of carbon, to reduce various metals in the manufacturing process. Unlike most elements, humans have been aware of carbon since ancient times. It occurs naturally as 3 isotopes – carbon 12, 13 and 14. It is tetravalent, having four electrons available to form covalent bonds. It is sometimes called the “king” of the periodic table.Ĭarbon is a non-metal, found in nature as graphite, diamond or fullerenes. Carbon has some similarities to the metalloid silicon, but silicon cannot be the basis for life like carbon can.
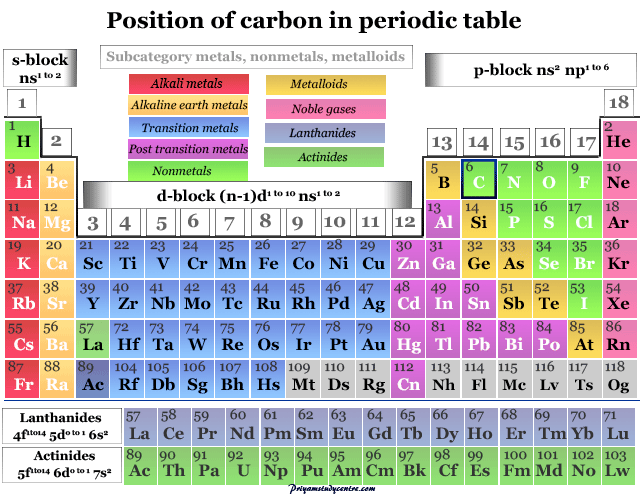
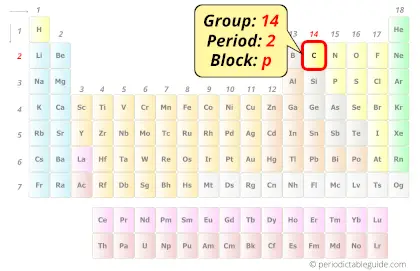
The elements silicon, germanium, tin and lead are also in group 14. It is believed the word carbon comes from the Latin word ‘carbo’ which means coal.Ĭarbon, with atomic number 6 and symbol C, resides in group 14 of the periodic table, to the right of boron and the left of nitrogen.The term graphite has Greek origin and is based on their term for ‘to write’.There is a wax layer on top of the carbon that disappears with pressure, transferring the carbon to the other page. Carbon copy paper is actually made with carbon.These allotropes include diamond, graphite, and graphene. Carbon is one of the few elements that can form stable covalent bonds with itself, and as a result, it can exist in many different forms, known as allotropes.Diamond is the hardest known natural substance on Earth, while graphite is one of the softest.Carbon is a key component of steel, which is one of the most widely used materials in the world.The human body is about 18% carbon by mass.Diamond is formed under great pressure deep in the earth’s crust, and are usually mined from ancient volcanic “pipes.”.The name is derived from the Latin “carbo” meaning charcoal.All other elements’ atomic weights are measured relative to carbon-12.Carbon is the 4th most abundant element in the universe but only the 15th most abundant element in the earth’s crust.Over 10 million compounds that we know of so far can be formed by carbon, more than any other element.All life as we know it is based on carbon.Carbon nanotubes are one of the strongest materials in the world and have been proposed as a possible building material for some wild inventions such as a space elevator.Measuring amounts of the radioactive isotope Carbon-14 can help us to determine the age of things such as archaeological artifacts and ancient documents.Find out more facts about this amazing element below. Diamond, graphite and charcoal are all forms of carbon. It is also one of the most important elements on the periodic table and forms the basis for all life as we know it! All of organic chemistry is based on carbon. And don’t forget to put the unit g/mol to your final calculated molar mass.Found in many forms, the element carbon is one of the most common elements in the universe.First solve the brackets, then multiplications and at last do the final addition. Always follow the calculation order to avoid any mistakes in calculation.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole.I hope you have understood the short and simple calculation for finding the molar mass of CO2. Hence the Molar mass of CO2 is 44.009 g/mol. So, Molar mass of CO2 = Molar mass of 1 Carbon (C) atom + Molar mass of 2 Oxygen (O) atoms. You can see that in CO2, there is 1 Carbon atom and 2 Oxygen atoms. Now, to calculate the molar mass of CO2, you just have to add the molar mass of all the individual atoms that are present in CO2.
